Recommendations to assure the quality, safety and efficacy of poliomyelitis vaccines (inactivated), Annex 3, TRS No 993

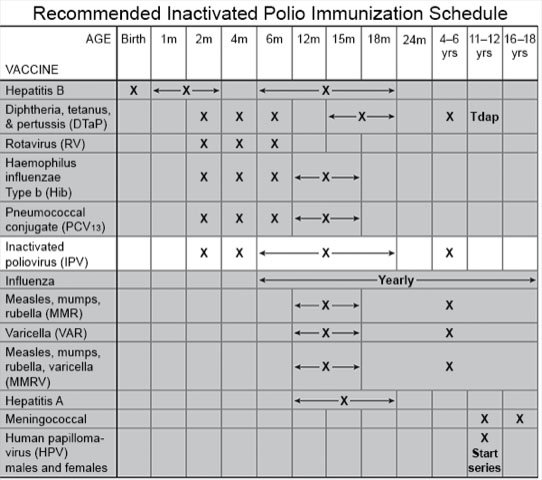
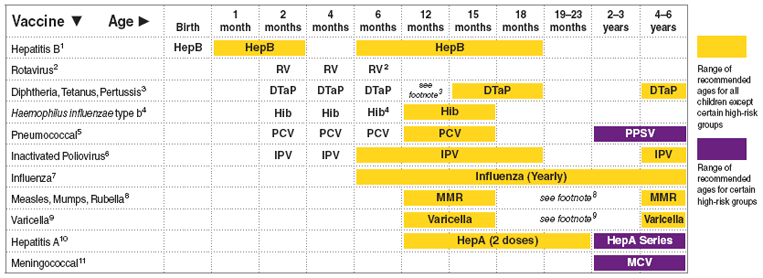
Poliomyelitis Prevention in the United States: New Recommendations for Routine Childhood Vaccination Place Greater Reliance on Inactivated Poliovirus Vaccine | Pediatric Annals

Years in the making: the World Health Organization prequalifies type 2 novel oral polio vaccine | PATH
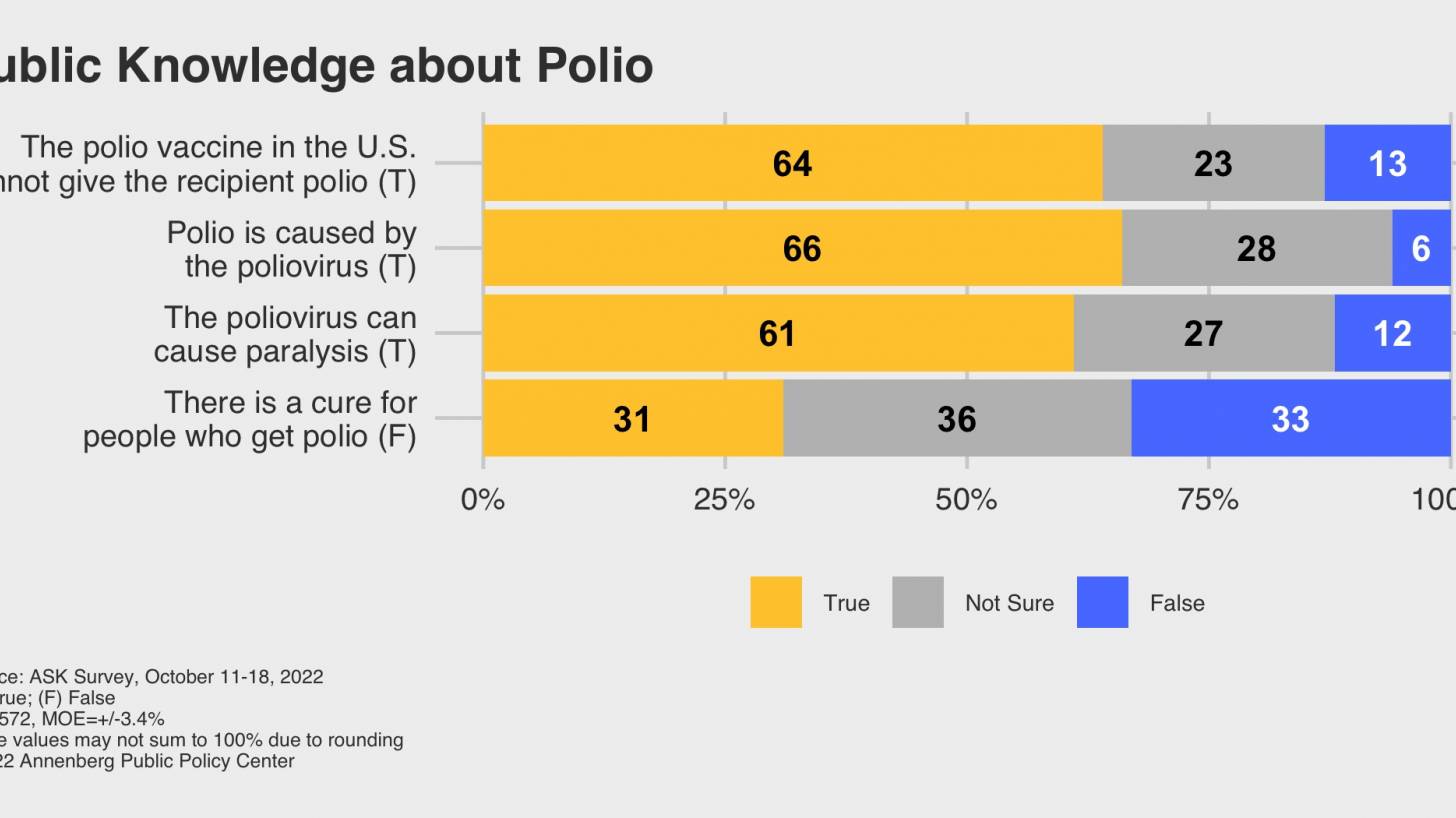
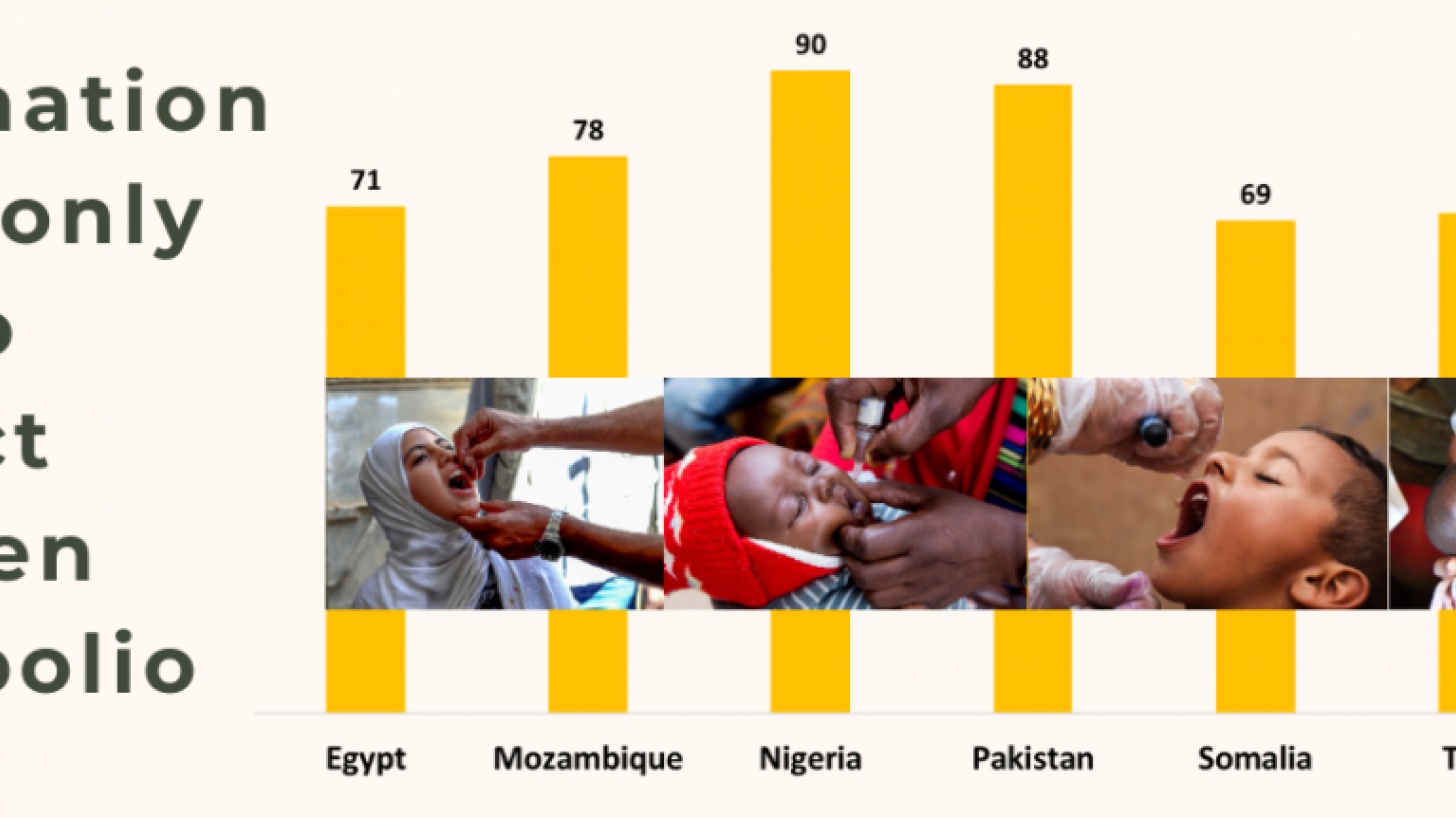
Strengthening polio vaccine demand in Ghana: Understanding the factors influencing uptake of the vaccine and the effectiveness of different message frames | PLOS ONE
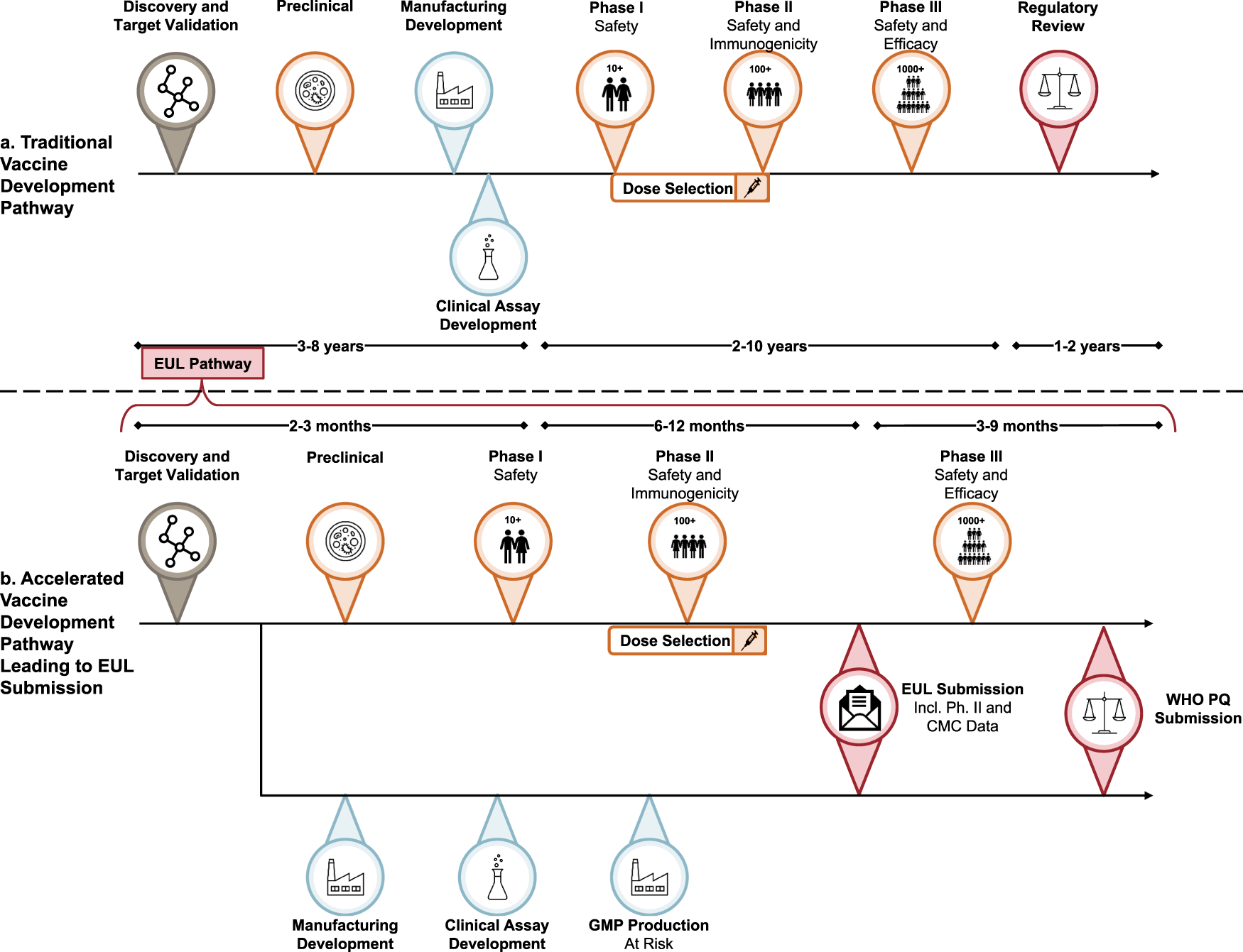
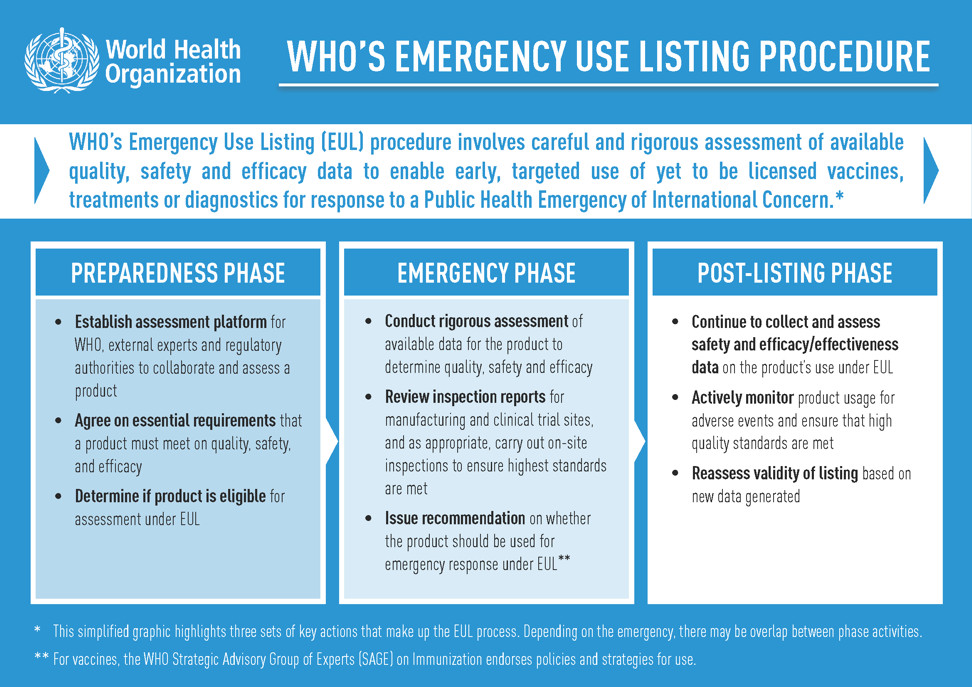
Recommendations for acceleration of vaccine development and emergency use filings for COVID-19 leveraging lessons from the novel oral polio vaccine | npj Vaccines