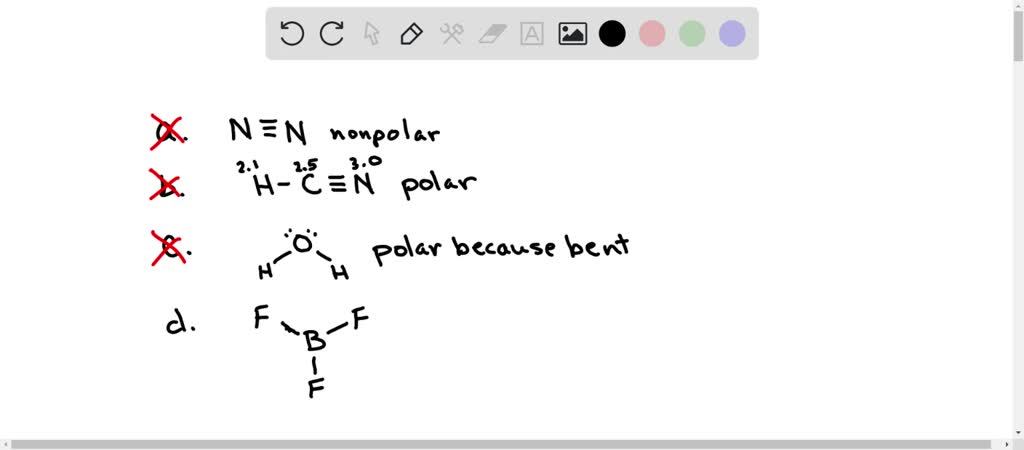
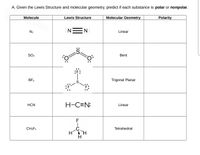
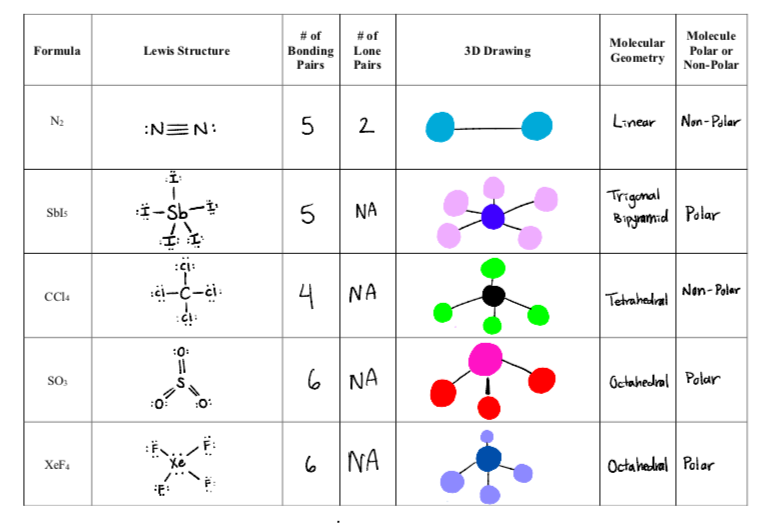
Is N2 Polar or Non-polar? (Nitrogen Gas) | Is N2 Polar or Non-polar? ( Nitrogen Gas) N2 is a chemical formula for Nitrogen gas and in today's video, we help you determine if

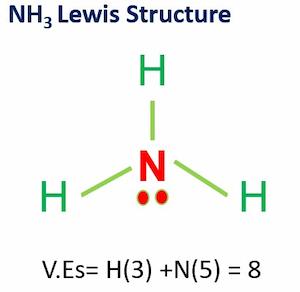
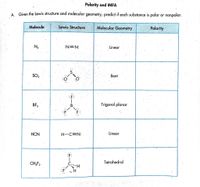
Which statement below best describes the Lewis structure for NCl3? a. Nitrogen has two single bonds, one double bond, and no lone pairs of electrons. One chlorine has one double bond and

Is N2 Polar or Non-polar? (Nitrogen Gas) | Is N2 Polar or Non-polar? ( Nitrogen Gas) N2 is a chemical formula for Nitrogen gas and in today's video, we help you determine if

N3- lewis structure, molecular geometry, hybridization, bond angle | Molecular geometry, Molecular, Electron configuration

N2 intermolecular forces - What types of Intermolecular Force is present in N2 molecule? along with the explanation of London Dispersion Forces and FAQs