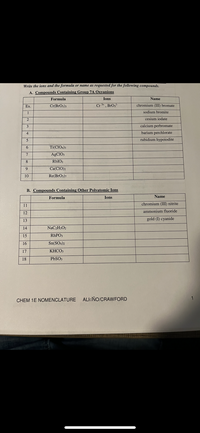
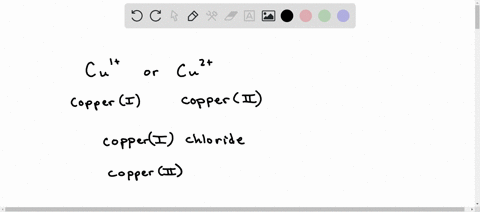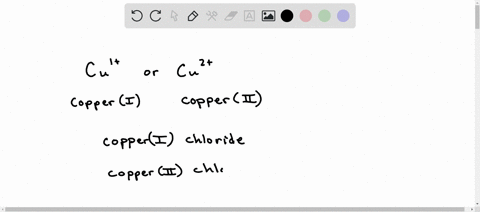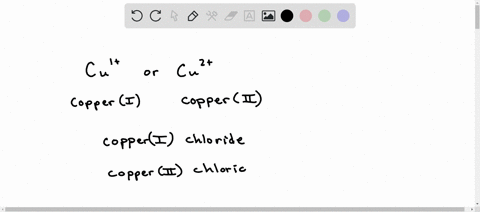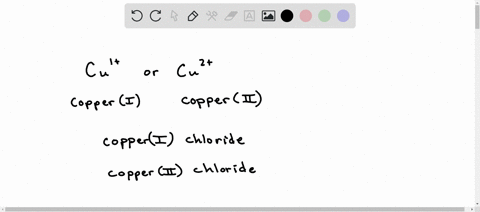
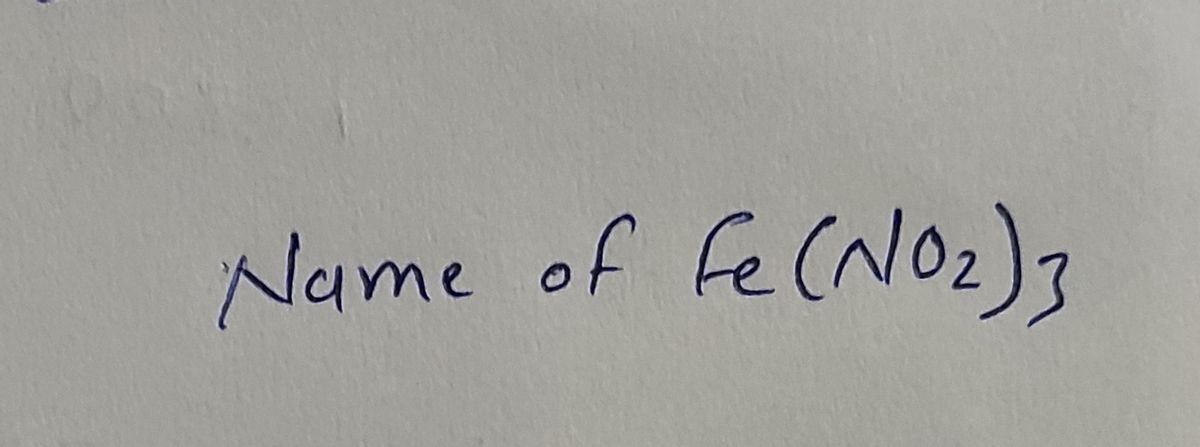Complete the following table below by writing the correct formula for the compound formed between each combination of cation and anion. Then write the correct chemical name for each. CO3 2? PO4
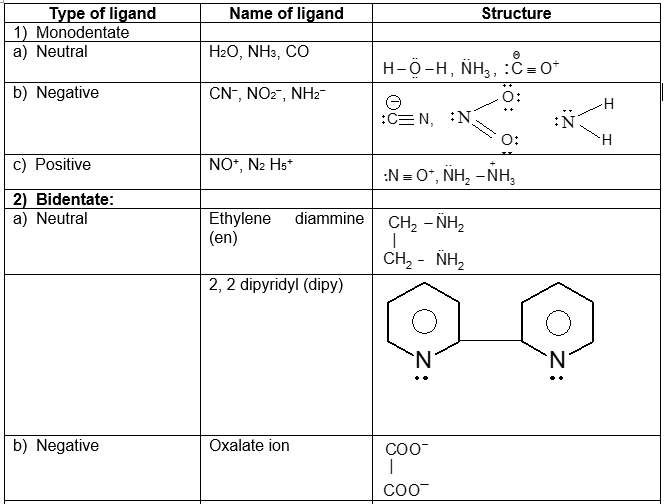
![SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ... SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...](https://cdn.numerade.com/ask_previews/3ac08f8-e66f-dd-b33c-3c46c11b4d2.gif)
SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...

Using Chemical Formulas 7-3 Beaker Breaker 1.Name the following compounds: CaCl 2 = ______ P 3 O 6 = ______ FeCl 2 = H 2 SO. - ppt download

Do Now Name the following compounds – KCl – Na 2 O – CuCl 3 Write the formulas for the following compounds – Magnesium chloride – Potassium sulfate – Tin. - ppt download

Reversible Extrusion and Uptake of HCl Molecules by Crystalline Solids Involving Coordination Bond Cleavage and Formation | Journal of the American Chemical Society