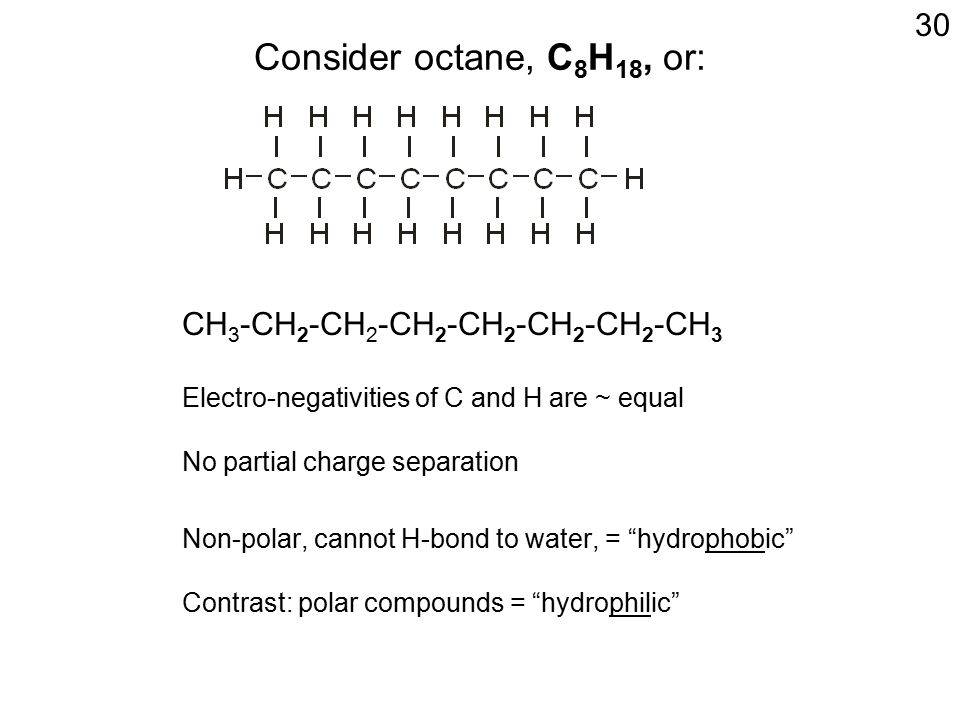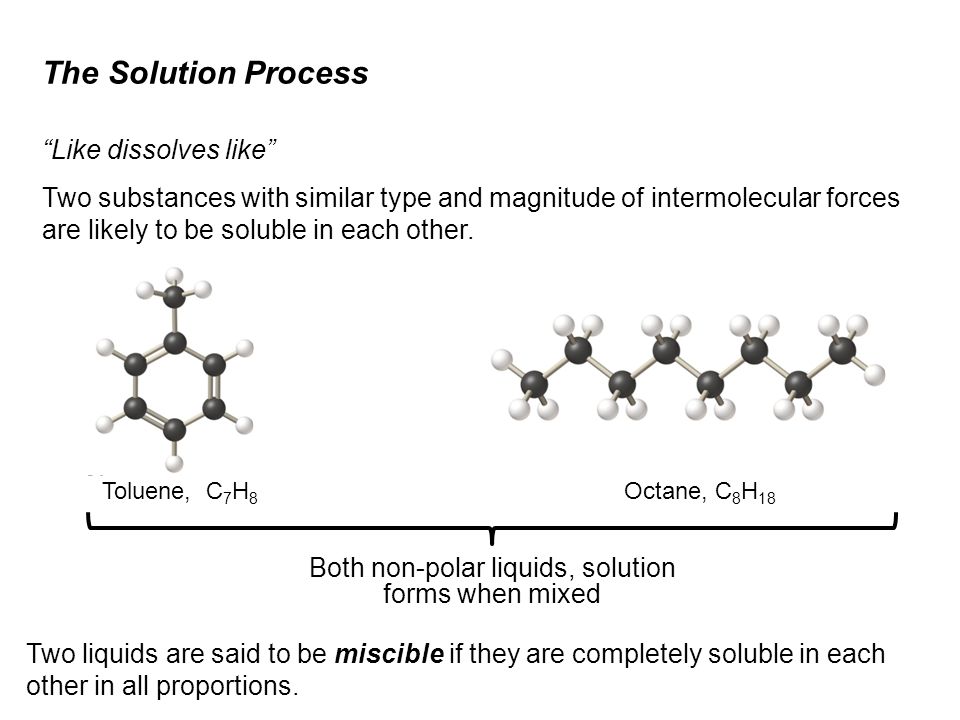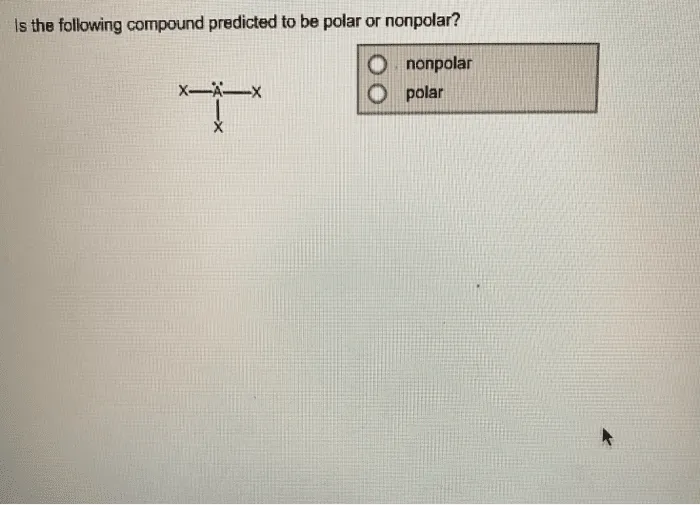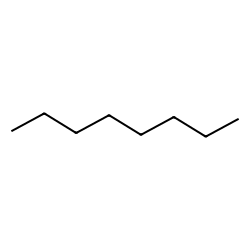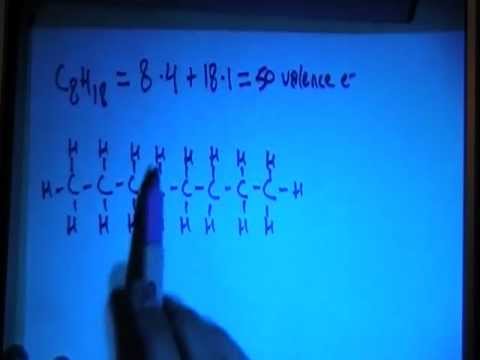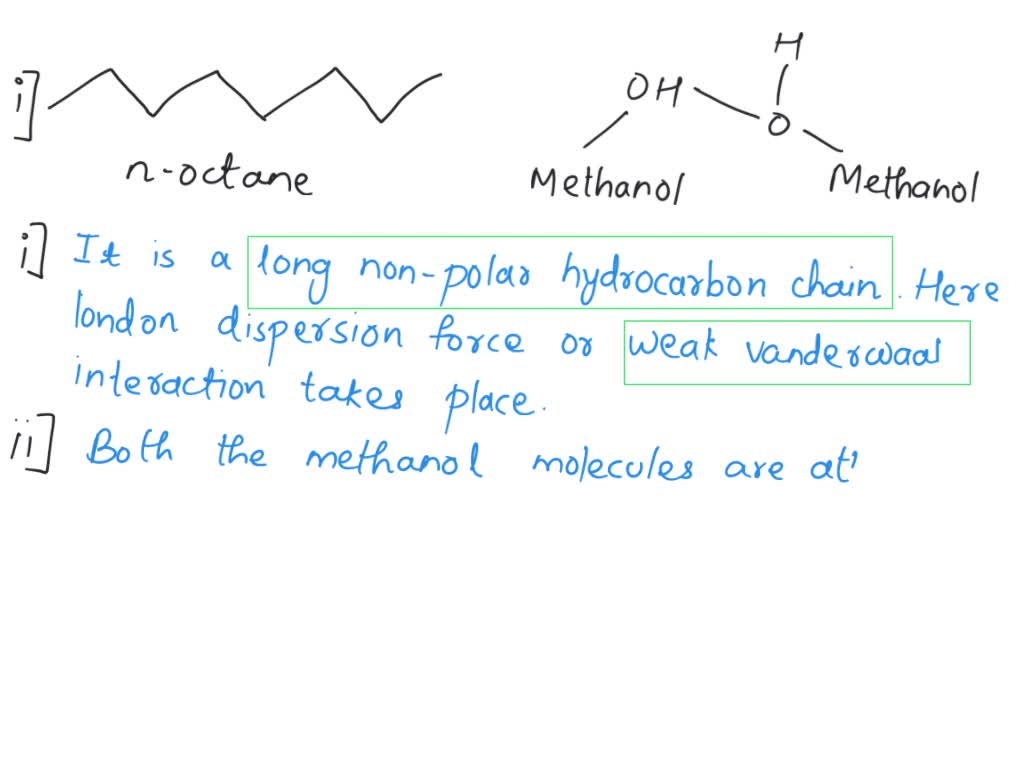
SOLVED: For n-octane (C8H18) and methanol (CH3OH), determine the predominant type of intermolecular attractions for the pure liquids (dispersion, permanent dipole, or hydrogen bonding). Predict whether these intermolecular attractions will be strong
Molecular structure of type II kerogen fragment (left), nonpolar oil... | Download Scientific Diagram

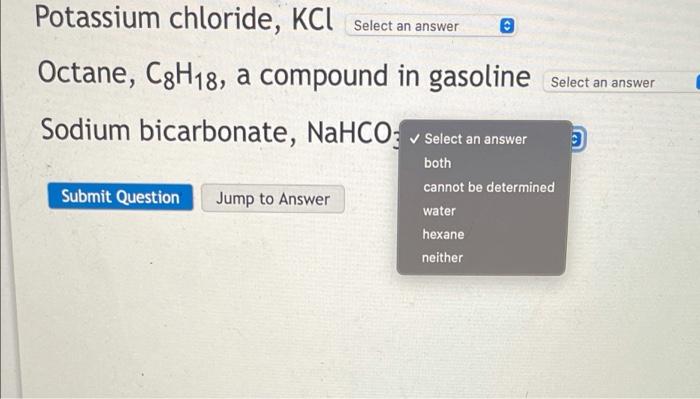
SOLVED: Water is a polar solvent and hexane is a non-polar solvent. Determine which solvent each of the following is most likely to be soluble in. Potassium chloride, KCl Octane, C8H18, a